Fibrosis Drug Discovery Services
Supporting anti-fibrotic therapeutics from discovery to clinic

How can you improve translational success in fibrosis drug discovery?
Despite promising preclinical findings, many fibrosis drug candidates fail in clinical development due to the limited predictive value of traditional models. A recurring roadblock to successful clinical trials for drugs that target fibrosis is translating positive results in animal models into human disease.
Fibrosis arises from complex interactions between fibroblasts, epithelial cells, immune cells, and the extracellular matrix across multiple organs. Capturing these mechanisms is critical for understanding therapeutic potential.
At Concept Life Sciences, we help bridge this translational gap by crafting innovative customized assays. Using human in vitro cell-based assays, immune-stromal co-cultures, 3D multicellular models, and spatial tissue profiling, we generate mechanistic insights and accurately quantify the effects of candidate drugs that target fibrotic processes, so that you can develop your anti-fibrotic drug programs with confidence.
We support research across multiple fibrosis indications:
- Pulmonary fibrosis and interstitial lung disease
- Liver fibrosis and metabolic dysfunction-associated steatohepatitis (MASH)
- Kidney fibrosis
- Dermal fibrosis and pathological scarring
- Endometriosis-associated fibrosis
Model and Cell Types:
- Lung fibroblasts
- Alveolar epithelial cells (AECII)
- Airway epithelial cells
- Macrophages
Representative Fibrosis Assays:
- TGF-β–induced fibroblast activation
- Myofibroblast differentiation (α-SMA)
- Collagen I/III deposition assays
- EMT assays
- ECM remodeling
- Fibroblast–macrophage co-culture
Model and Cell Types:
- Hepatic stellate cells
- Kupffer cells
- Hepatocytes
Representative Fibrosis Assays:
- Stellate cell activation
- Lipotoxicity-driven fibrosis
- Collagen secretion/deposition
- Cytokine-induced fibrosis
- HSC–macrophage co-culture
Model and Cell Types:
- Renal fibroblasts
- Proximal tubule epithelial cells
- Podocytes
Representative Fibrosis Assays:
- EMT assays
- TGF-β fibroblast activation
- Tubulointerstitial fibrosis models
- Collagen deposition/ECM assays
- Epithelial–stromal co-culture
Model and Cell Types:
- Cardiac fibroblasts
- Cardiomyocytes
- Cardiac macrophages
Representative Fibrosis Assays:
- Myofibroblast conversion
- TGF-β cardiac fibrosis
- ECM protein assays
- Fibroblast–cardiomyocyte co-culture
- Contractility and fibrosis models
Model and Cell Types:
- Dermal fibroblasts
- Keratinocytes
Representative Fibrosis Assays:
- Fibroblast activation
- Scleroderma-mimetic fibrosis assays
- Wound healing fibrosis models
- EMT assays
- Fibroblast–keratinocyte co-culture
Model and Cell Types:
- Intestinal fibroblasts
- Epithelial cells
- Mucosal immune cells
Representative Fibrosis Assays:
- Fibroblast activation
- Cytokine-driven remodeling
- Collagen deposition
- Epithelial–fibroblast co-culture
Model and Cell Types:
- Endometrial stromal cells (ESCs)
- Endometriotic lesion–derived fibroblasts
- Epithelial endometrial cells
- Peritoneal macrophages
Representative Fibrosis Assays:
- Lesion-associated fibroblast activation (α-SMA, COL1A1)
- ESC decidualization and fibrosis modulation
- Cytokine-driven ECM buildup
- Macrophage co-culture to model inflammatory fibrosis
- Collagen deposition and ECM remodeling
Model and Cell Types:
- Pancreatic stellate cells
- Pancreatic fibroblasts
Representative Fibrosis Assays:
- Stellate cell activation
- ECM fibrosis models
- Tumor–stroma interaction assays
- Stellate–cancer co-culture
Model and Cell Types:
- RPE cells
- Conjunctival fibroblasts
Representative Fibrosis Assays:
- EMT-driven fibrosis
- TGF-β ECM deposition
- Post-surgical scarring models
Model and Cell Types:
- Mesenchymal stromal cells (MSCs)
- Primary fibroblasts (various tissues)
Representative Fibrosis Assays:
- Pan-fibrotic screening
- Collagen I/III quantification
- High-content imaging of fibrosis markers
- Mechanism-of-action phenotypic assays
Fibrosis services
Driving your fibrosis drug discovery goals to success
Excessive deposition of connective tissue during wound healing can lead to progressive and irreversible damage across multiple organs. As a result, diseases driven by fibrotic pathology—whether in the lung, liver, kidney, skin, or other tissues—are now recognised as major contributors to global morbidity and mortality. Although new drug candidates are emerging, a key challenge remains: how can we adapt cell‑based assays to more accurately model tissue‑specific fibrotic mechanisms and overcome the limitations inherent in animal models?
Concept Life Sciences employs mesenchymal and epithelial cell culture systems derived from a range of relevant tissues to interrogate activation, differentiation and collagen deposition in vitro. This flexibility enables us to mimic fibrosis pathways in organ‑specific contexts. Therapeutics designed to modulate pro‑fibrotic responses can be added at multiple stages of the assays to target distinct phases of fibrosis progression. To further increase physiological relevance, co‑culture systems incorporating accessory cells such as macrophages can be introduced, allowing us to explore immune‑fibroblast crosstalk and better emulate the complexity of tissue microenvironments.
We offer a suite of high-quality cell-based assays to investigate candidate drugs in:
- Mesenchymal cells
- Tissue fibrosis
- Accessory cells in fibrosis including macrophages and T cells
- Multicellular MASH models
- Epithelial - mesenchymal transition
Mesenchymal cells
The differentiation of tissue resident mesenchymal cells, such as fibroblasts or pericytes, into myofibroblasts is a key process for effective wound healing. However, it is difficult to assess direct effects of a candidate therapeutic in vivo and animal cell culture does not always translate to clinical success.
Mesenchymal cell differentiation
To investigate mesenchymal cell differentiation / activation, we culture human fibroblasts and measure changes in viability and gene expression of known activation biomarkers utilizing TGF-β, a recognized pro-fibrotic factor, as a positive control.

Mesenchymal cell phenotyping
High content imaging (HCI) can be used to analyze multiple cellular parameters including collagen production and secretion, cell viability and cell morphology. HCI is suitable for large scale screening studies and provides a robust assessment of phenotypic changes exhibited by mesenchymal cells in response to different treatments.

Pro-collagen
An additional approach for analyzing collagen type I synthesis is by measuring the secretion of precursor procollagen peptides. Procollagen peptides facilitate the winding of procollagen molecules into a triple-helix and are then cleaved during secretion. The amount of free pro-peptide directly correlates with the synthesis of collagen and can be measured using antibody-based assays. This approach is particularly suited to early screening and efficacy studies
High‑resolution spatial profiling of fibrotic tissues
Precise characterization of therapeutic effects within intact tissue architecture is essential for understanding mechanistic activity in fibrotic disease. Our Spatial Biology group employs multiplex immunofluorescence, RNA spatial profiling, and high‑resolution histological imaging to quantify collagen deposition, fibroblast activation states, immune–stromal interactions, and regional heterogeneity within fibrotic lesions. These spatially resolved datasets allow us to track how candidate therapeutics modulate microenvironmental niches, providing a robust framework for mechanistic interpretation and translational prediction beyond conventional in vitro or bulk tissue assays.
Accessory cells in fibrosis
Investigating the effects of candidate drugs on human cells is critical to avoid inaccuracies found when translating animal models to human clinical trials. However, selecting disease appropriate cells and assay systems requires expert advice. We offer primary human cell mono-culture assays to confirm potency, efficacy, mode of action and toxicity of accessory cells in vitro. Assay complexity can be increased by co-culturing target cells with mesenchymal cells.
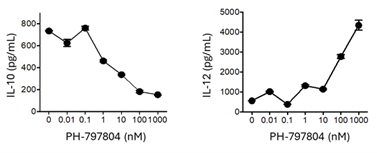
Macrophage assays
Macrophages are the architects of tissue repair, supporting mesenchymal differentiation, resolving inflammation and promoting tissue repair. However, aberrant macrophage activation leads to fibrosis and immune mediated pathology. Human monocyte derived macrophages can be polarized to disease associated phenotypes and the effect of candidate therapeutics on phenotype and function determined.
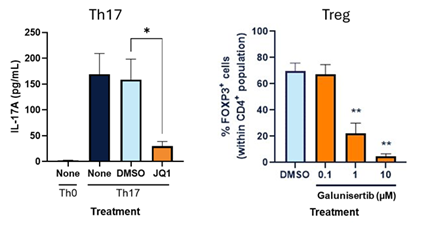
T cell assays
T cells influence fibroblast and macrophage activation through the release of pro- and anti- inflammatory cytokines. Therefore, modifying influential T cell subsets is an important strategy in drug development for fibrotic disease.
We culture primary human T cells with candidate therapeutics to determine whether they alter the inflammatory response. T cells can be polarized to generate disease relevant phenotypes or co-cultured with other cell types of interest.
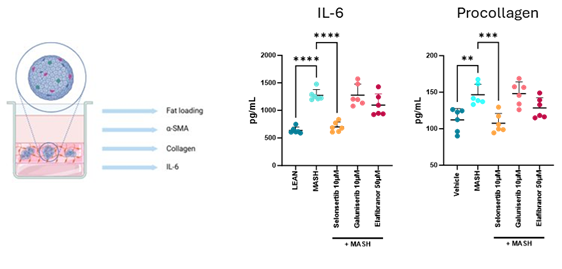
Multicellular 3D MASH models
A challenge facing preclinical research is providing assays that imitate the complexity of disease in vivo without relying on animal models. We offer a metabolic associated steatohepatitis (MASH) model that adopts a three-dimensional culture of hepatocytes, stellate cells and macrophages. Cells can be stimulated by lipids in the presence of candidate drugs and changes in cytokine production or pro-collagen determined.
Epithelial to mesenchymal transition
Epithelial to mesenchymal transition (EMT) is a physiological process integral to organogenesis, tissue development, and wound healing. However, it also plays a pathological role in tumor progression, metastasis, and fibrosis. Assays that model epithelial to mesenchymal transition are difficult to find. To investigate EMT during fibrosis, we measure changes in collagen type I and alpha-smooth muscle actin within a scratch wound assay. The assay models the conversion of epithelial cells into fibroblasts and the subsequent wound healing processes, following tissue damage.
Macrophages are pivotal in modulating EMT and a target for novel therapeutics targeting fibrosis. The scratch wound assay can be adapted to include macrophages within the epithelial cell culture supporting analysis of functional changes in co-cultures treated with candidate drugs targeting fibrosis.
