Microsome and S9 Stability

In vitro metabolism assay
In early drug discovery, compounds can show strong in vitro potency yet fail in vivo because of rapid drug metabolism and insufficient exposure. Early metabolic stability testing helps teams identify clearance risk sooner, prioritize series, and focus lead optimization on the changes most likely to improve in vivo performance.
A microsomal stability assay, with optional S9 fractions, provides a rapid, practical way to estimate metabolic turnover and generate decision-ready stability parameters as part of ADMET screening.
Key benefits
The primary value of microsome and S9 stability testing is improving decision quality, helping teams identify which compounds to progress and how to optimize those that require improvement.
Species flexibility for translational confidence: Studies can be run using pooled liver microsomes or S9 fractions from human, mouse, rat, dog, and monkey, supporting series ranking and early species strategy.
Broader metabolic coverage when it matters: Microsomes are ideal for CYP-driven Phase I stability, while S9 adds additional enzymatic pathways and can capture Phase II contributions (with UDPGA), helping teams avoid missing clearance mechanisms.
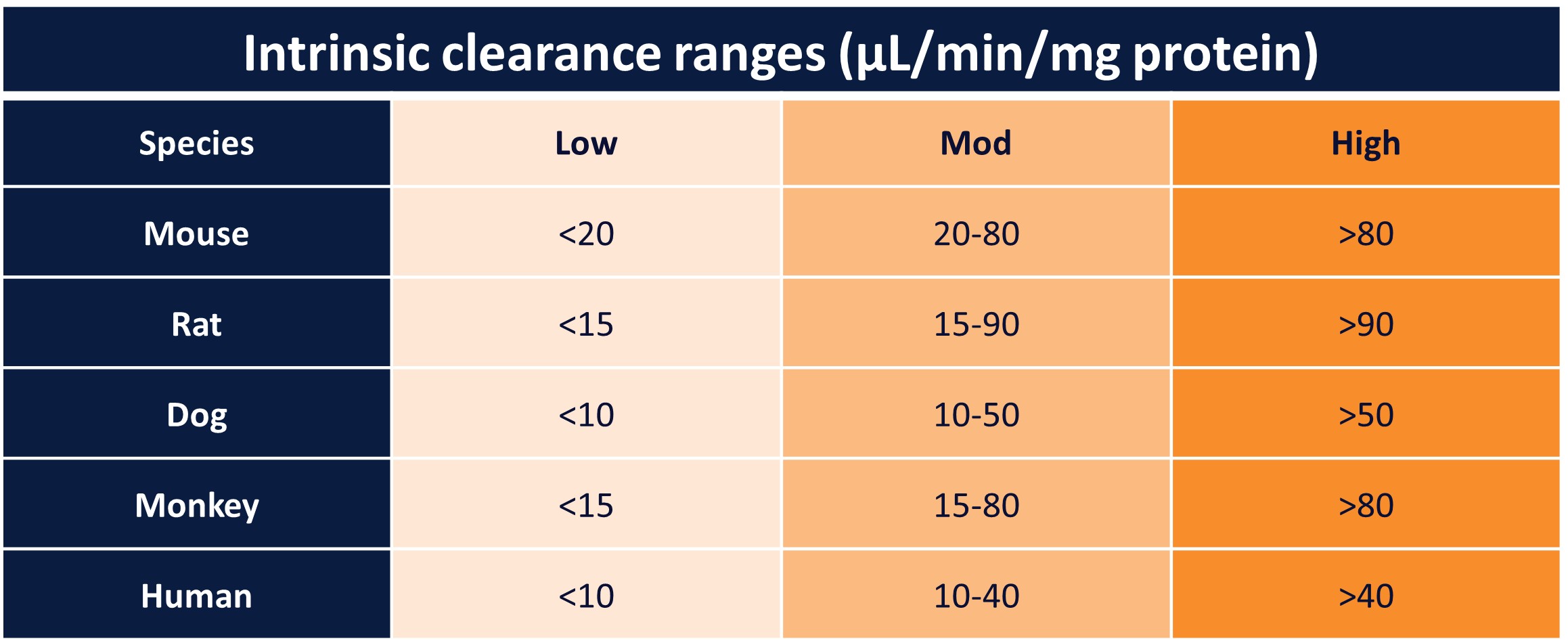
Robust, reproducible readouts: outputs include intrinsic clearance (CLint), half-life (t½ ), and elimination rate constant (k), parameters commonly used to compare compounds and estimate clearance risk.
Supports lead optimization: Stability results help identify when metabolism is likely to limit exposure and guide structural refinement (e.g., reducing clearance or addressing metabolic “hot spots”) rather than relying on trial-and-error iteration.
Cost-effective: High-throughput format with LC-MS/MS quantitation supports rapid turnarounds for iterative medicinal chemistry cycles.
Protocol
Pooled liver microsomes or S9 fractions are incubated with test compound at controlled temperature in the presence of appropriate cofactors to initiate biotransformation. Samples are collected at defined time points, reactions are quenched, and proteins are precipitated. The remaining parent compound is quantified by LC-MS/MS to determine compound depletion over time and calculate metabolic stability parameters.
Microsomes
Microsomes are diluted to an assay concentration of 1 mg protein/mL and pre-incubated with NADPH cofactor for 10 minutes at 37°C. Biotransformation is initiated by the addition of the test compound and mixing. Samples are typically collected at 0, 5, 10, 15, 25, and 35 minutes, or at client-defined time points.
S9
S9 fractions are diluted to an assay concentration of 1 mg protein/mL in the presence of alamethicin (in MeOH) and MgCl2 and pre-incubated with NADPH and UDPGA cofactors for 10 minutes at 37°C. Biotransformation is initiated by the addition of the test compound and mixing. Samples are collected at standard or client-defined time points, consistent with microsomal stability studies.
Reaction Quench
The final solvent concentrations in the incubations are 0.99% methanol and 0.01% DMSO. At each specific time-point, a sample aliquot (25µL) is removed from the test incubation mixture and immediately combined into a cassette of up to four compounds, in 300µL ice cold methanol containing internal standard and mixing to stop the reaction.
After the final time-point, the quenched samples are centrifuged (4000 rpm at 4°C for 30 min) to precipitate the protein.
Study Requirements
Clients typically provide:
- Compound identifier and molecular formula
- Test material: 25µL of 10 mM solution in DMSO or 0.5 mg solid
Test System and Conditions
- Species available: human, rat, mouse, dog, and monkey
- Typical incubation concentration: 1µM
- Format: 96-well plate, shaking incubator at 37°C
Controls and Quantitation
Each assay includes standard positive controls (dextromethorphan, diazepam, midazolam, and phenacetin) to ensure assay performance. Supernatants are analyzed by LC-MS/MS to quantify the parent compound remaining at each time point.
Data analysis and results
For each test compound injection:
Response ratio = Test compound peak area / Internal standard peak area
The natural logarithm of the response ratio is plotted versus time. The slope of the semi-log plot provides the elimination rate constant (k), from which we calculate:
- Half-life (t½) (t½ = ln2 / k)
- Intrinsic clearance (Clint)
These parameters enable comparison across compounds, identification of clearance risk, and ranking within compound series to support progression decisions.


Conclusions
Microsome and S9 stability assays provide an early, reliable assessment of metabolic clearance and pathway risk. By enabling rapid compound ranking, identifying clearance liabilities, and guiding targeted optimization, these studies help teams focus resources on compounds with the greatest likelihood of achieving sufficient in vivo exposure.
