Kinetic Solubility

Drug solubility solutions for drug development
At Concept Life Sciences, solubility is more than a measurement—it’s a critical factor that influences a drug’s success from formulation through therapeutic efficacy. Poor solubility can lead to low absorption, compromised bioavailability, and inaccurate assay results, ultimately increasing development time and cost. These challenges often correlate with high plasma protein binding, slow tissue distribution, and drug-drug interaction risks.
Advance your program with reliable drug solubility testing that reduces developability risk and supports confident formulation and development decisions. Our CRO services include:
- Kinetic and thermodynamic solubility using the shake-flask method for rapid screening or precise equilibrium measurements.
- Biorelevant media testing in Simulated Gastric Fluid (SGF), Fasted State Simulated Intestinal Fluid (FaSSIF), and Fed State Simulated Intestinal Fluid (FeSSIF) to predict in vivo performance accurately.
By combining robust solubility data with physiologically relevant conditions, we help you make informed decisions that accelerate drug development and reduce risk.
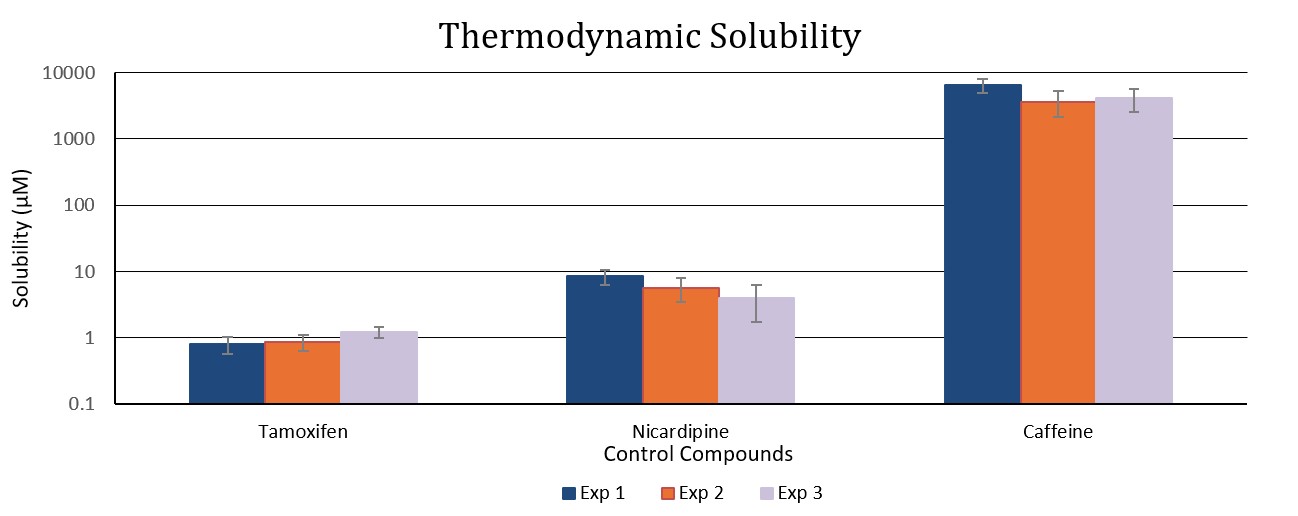
Thermodynamic solubility
Thermodynamic solubility determination is performed using dry solid material, where particle size and crystal morphology can significantly influence the amount solubilized at equilibrium. This method provides robust and accurate data that are highly relevant for drug development, supporting decisions in areas such as pharmacokinetic (PK) studies and enabling strategies commonly applied during the drug development stages to address poor solubility.
Deliverable: Aqueous solubility concentration expressed in micromolar (μM).
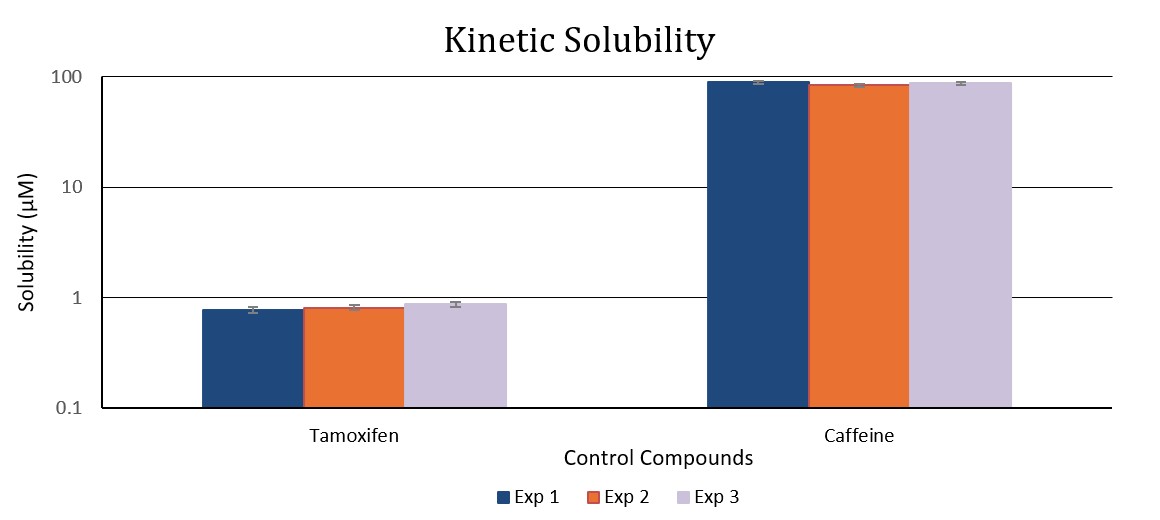
Kinetic solubility
Kinetic solubility tests are quick, high-throughput assays typically performed in the early phases of drug discovery to rapidly assess compound solubility under screening-like conditions. The solubility of a compound can be significantly influenced by the presence of dimethyl sulfoxide (DMSO), which is commonly used as a storage solvent during early drug development.
Our kinetic solubility determination uses a DMSO stock solution to replicate conditions encountered in early in vitro biological assays, where test solutions typically contain trace amounts of DMSO. This approach ensures that solubility measurements are both relevant and representative of real-world screening environments, supporting efficient decision-making in lead optimization.
Deliverable: Aqueous solubility concentration expressed in micromolar (μM).
Key benefits
Comprehensive expertise
- Full spectrum of solubility assays: from rapid kinetic screening to precise thermodynamic measurements.
- Accurate, reproducible data tailored to your development stage.
State-of-the-art facilities
- Advanced instrumentation and validated protocols.
- High-quality results aligned with regulatory standards to support critical decisions.
Integrated drug development support
- End-to-end solutions that fit seamlessly into your development workflow.
- Expertise that accelerates progress from early screening to late-stage optimization.
Customizable solutions
- Bespoke assay packages designed for your compound’s unique challenges.
- Flexible options for early discovery or advanced optimization needs.
Speed without compromise
- Rapid turnaround times without sacrificing quality.
- Reliable data delivered when you need it most—reducing risk and shortening timelines.
Protocol
Customer provides
- Compound identifier and molecular formula.
- Thermodynamic: 2 x 1 mg solid.
- Kinetic: 0.5 mg solid or 50 μL 10mM DMSO stock.
Format
- 1.5mL glass vials. Aqueous pH7.4 phosphate buffer 0.1M (or customer specific). Orbital shaking incubator at 25ºC.
Buffers
- pH7.4 phosphate buffer (0.1 M)
- Multiple pH values available upon request
- Biorelevant media: FaSSIF, FeSSIF, and simulated gastric fluids (SFG)
Procedure

For thermodynamic experiments, weigh 1 mg of the test compound. For kinetic experiments, pipette 10 μL of a 10 mM DMSO stock solution of the compound. Add the compound to 1000 μL of buffer for thermodynamic studies or 990 μL of buffer for kinetic studies in a clean glass vial. Seal the vial and place it on an orbital mixer, shaking for 24 hours to reach equilibrium in thermodynamic experiments or for 1 hour in kinetic experiments.
After mixing, centrifuge the solution at 3000 rpm at room temperature for 20 minutes. Carefully transfer the supernatant to a second vial and centrifuge again under the same conditions. From the final supernatant, prepare two dilutions—a high and a low concentration. These samples will be used for LC-MS/MS quantitation against a standard curve. Include an internal standard in the final dilution for analysis.
Controls
Tamoxifen is used as the low solubility control and caffeine as the high solubility control, providing reference points to bracket the assay range and verify assay performance.
Quantitation
LC-MS analysis using a 4-point standard curve prepared from a 10 mM DMSO stock.
Response ratio
Test peak area / Internal standard peak area.
- Compound identifier and molecular formula.
- Thermodynamic: 2 x 0.5mg solid.
- Kinetic: 0.5mg solid or 50μl 10mM DMSO stock.
Data analysis and results
For each test compound injection:
Response Ratio =Test peak area / Internal standard peak area.
Test compound response ratio for incubated high and low concentrations are quantified against the standard curve to produce an aqueous solubility concentration (µM).
Conclusions
From kinetic solubility for rapid screening to thermodynamic solubility for precise equilibrium measurements, we deliver comprehensive solubility testing solutions that accelerate drug development. Our state-of-the-art facilities, validated protocols, and expert scientists ensure accurate, reproducible data that meets regulatory standards.
We offer customizable assay packages tailored to your compound’s unique challenges—whether for early-stage screening, formulation support, or late-stage optimization. With fast turnaround times and rigorous quality control, we help you reduce risk, improve decision-making, and shorten timelines.
